 |
Need a New Laboratory Water System?
We have many to choose from. We can save you thousands on
Complete Systems and replacement filters for most brands. |
Hard Water and Water Softening
 |
Also See :
How to select the best water softener
Comprehensive Water Softener Calculator
NEW : Chemical Free Water Softener
What impurities will softeners remove?
Water softeners will remove nearly all the calcium and magnesium from the raw water during the softening process. Softeners will also remove up to 10 ppm of iron and manganese. Water supplies with high levels of iron and manganese 'greater than 10 ppm' may need pretreatment to prolong the lifespan of a water softener.
What makes water hard ?
Groundwater dissolves rocks and minerals releasing calcium and magnesium ions that cause water to be hard. These dissolved ions give hard water its characteristics.
The Term 'hard water' comes from the type of deposits that are left behind when the water evaporates leaving behind residue. Calcium and magnesium leave behind a hard residue that is difficult to wipe off. This is know as an insoluble salt. Sodium on the other hand leaves a soft residue that easily wipes off. This is known as a soluble salt.
Water Hardness Map
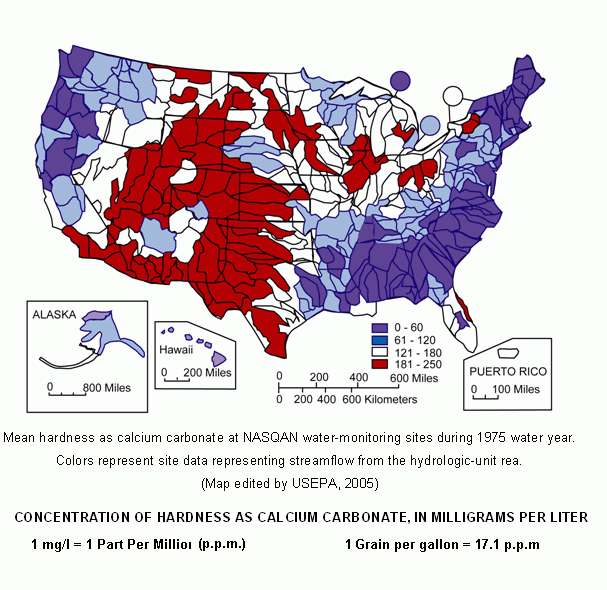
(Click for larger image)
How is hardness measured?
Water hardness ordinarily is expressed in grains of hardness per gallon 'gpg' of water.
Water impurities can also be measured in either parts per million 'ppm' or milligrams per liter 'mg/l'.
One gpg is equal to 17 ppm or 17.1 mg/l. Table 1 shows how hardness is classified.
Table 1. Hardness Classification
----------------------------------------------------------
Level of Hardness in grains 'gpg'
----------------------------------------------------------
below 1.0 soft
1.0 to 3.5 slightly hard
3.5 to 7.0 moderately hard
7.0 to 10.5 hard
above 10.5 very hard
----------------------------------------------------------
Problems caused by hard water
Hard water interferes with all types of cleaning tasks. Cleaning problems arise when the cleaning agents do not fully remove dirt and grime. Over time, clothes washed in hard water may look dingy and feel harsh and scratchy. White clothing continually washed in hard water will gradually show a grayish tinge. Dishes and glassware washed in dishwashers using hard water may be spotted when dry. Hard water causes films on glass shower doors, walls and bathtubs. Hair washed in hard water may feel sticky and look dull.
Regular soaps combine with dissolved calcium and magnesium to form soap curds or soap scum. Soap scum is difficult to remove from sinks and appliances.
Household appliance performance may be affected by hard water use. When heated, calcium carbonate and magnesium carbonate are removed from the water and produce a scale buildup in the hot water heater. A large scale buildup slows the heating process and requires more energy to heat water. Water heaters with large accumulations of mineral buildup will have shorter life spans. Scale deposits also corrode and plug plumbing fixtures and accumulate in other appliances affecting their performance.
The ion exchange process
Calcium 'Ca2_' and magnesium 'Mg2_' ions that cause water hardness can be removed fairly easily by using an ion exchange procedure. Water softeners are cation exchange devices. Cations refer to positively charged ions. Cation exchange involves the replacement of the hardness ions with a nonhardness ion. Water softeners usually use sodium 'Na_' as the exchange ion. Sodium ions are supplied from dissolved sodium chloride salt, also called brine. In the ion exchange process, sodium ions are used to coat an exchange medium in the softener. The exchange medium can be rural zeolites or synthetic resin beads that resemble wet sand.
As hard water passes through a softener, the calcium and magnesium trade places with sodium ions. Sodium ions are held loosely and are easily replaced by calcium and magnesium ions. During this process free sodium ions are released to the water.
Softening Process
NaZeolite _ Ca2 --> CaZeolite _ Na_
and
NaZeolite _ Mg2_ --> MgZeolite _ Na_
After softening a large quantity of hard water the exchange medium becomes coated with calcium and magnesium ions. When this occurs, the exchange medium must be recharged or regenerated 'Figure 1'. To recharge the softener with sodium ions, a softener is backflushed with a salt brine solution. During a backflush the brine solution replaces the calcium and magnesium ions on the exchange medium with sodium ions from the salt solution.
Recharging Process
NaC1 _ CaZeolite --> NaZeolite _ CaC1
and
NaC1 _ MgZeolite --> NaZeolite _ MgC1
The time between recharging cycles depends on the hardness of the water, the amount of water used, the size of the unit, and the capacity of the exchange media to remove hardness.
Water testing
Before buying any water treatment equipment, you should know what impurities are found in the water supply. Types and amounts of impurities in your water can be determined by a certified laboratory. The results of the water test will help determine if softening is needed. The water testing may reveal if other water treatment is required. If you obtain water from a private water supply, water testing is your responsibility. Water testing should be done on a regular basis. If a problem is suspected, test more often.
Community water supplies are monitored and treated to protect users from health threatening water impurities. Ask your supplier for a copy of the latest water test results. Hard water is considered a nuisance water problem. Hardness removal is not a necessity to protect your health. Water softening is popular because most people prefer softened water for bathing, cleaning and washing.
Health risks associated with softened water
During the softening process sodium is released from the exchange media into the output water. For every grain of hardness removed from water, 8 mg/1 'ppm' of sodium is added. People on restricted sodium intake diets should account for increased levels of sodium in softened water. Your family physician should be consulted. Sodium intake from softened water can be avoided by leaving one kitchen tap unsoftened for drinking and cooking.
Types of water softening equipment available
Water softeners are classified in five different categories:
How to select the right softener
1. Manual: There are several types of manual softeners. The operator opens and closes valves to control the frequency, rate and time length of backflushing or recharging.
2. Semi-automatic: The operator initiates only the recharging cycle. A button is pushed when the softener needs recharging and the unit will control and complete the recharging process.
3. Automatic: The automatic softener usually is equipped with a timer that automatically initiates the recharging cycle and every step in the process. The operator needs only to set the timer and add salt when needed. It is the most popular type of softener used.
4. Demand initiated regeneration 'DIR': All operations are initiated and performed automatically in response to the water use demand for softened water. DIR systems generally have two softening tanks and a brine tank. While one tank is softening the other tank is recharging.
5. Off-site regeneration 'generally rental units': A used softening tank is physically replaced with a recharged tank. Spent softening tanks are then recharged at a central location.
Operation and maintenance
Maintenance of water softeners is largely confined to restocking the salt supply for the brine solution. With manual and semiautomatic models, the owner will also have to start the recharging cycle. Salt can be purchased in the form of pellets, granules or blocks.
The brine tank may require periodic cleaning. The frequency of cleaning depends on the amount and purity of the salt used in the softening process. The brine valve and float assembly should also be checked and cleaned as often as needed.
The presence of excess iron or hydrogen sulfide can inhibit the effectiveness of a water softening unit. Installation of the proper presoftening treatment equipment may be required 'see the Iron and Manganese circular in the Household Water Treatment series'. Water test results will help make that determination. More frequent back-washing or reversing the normal flow of water through the treatment unit may be required to remove iron buildup.
Cost of water softeners and supplies
Retail prices for home water softeners may range from approximately $400 to $1200 depending on the size and type of softener. Softeners are rated by the total number of grains the unit can remove before being recharged. Cost of salt is approximately $5 to $7 per 40 pound bag depending on the form purchased.
Advantages of water softeners
Softeners offer:
1. cleaner, softer feeling clothes;
2. longer life of appliances including washing machine, dishwasher, and water heater;
3. less use of household cleaning products, such as detergents, as well as personal cleanliness products, like shampoo;
4. reduction of water spotting.
Disadvantages of water softeners
1. Softened water is not recommended for watering house plants, lawns and gardens due to its sodium content;
2. water used in recharging a water softener may over load or reduce the effectiveness of small septic or sewer systems;
3. there may be health risks from sodium intake;
4. softened water is not recommended for steam irons or evaporative coolers. The best choice for such appliances is distilled water or water from a Reverse Osmosis unit.
Alternatives to ion exchange units
Hard water problems can be reduced by using detergents that include water softening chemicals in their formulation. Some types of chemicals can be added to hard water to reduce the negative effect from calcium and magnesium. Chemical treatment for household water softening is recommended for low levels of hardness. Two types of chemicals used to soften water for home laundry are Sal Soda and Calgon.
Sal Soda combines with calcium and magnesium to form solid particles. These particles settle out with particles of dirt during washing. Use of precipitating additives such as Sal Soda may not fully clean your laundry because solid particles may cling to fabrics. Calgon softens water by combining with calcium and magnesium to form compounds that stay in solution. Use of nonprecipitating additives such as Calgon has a negative environmental effect because they have high phosphate content.
Magnetic Conditioning
Permanent magnetic water conditioning devices have been marketed based on a variety of claims regarding their effect on water hardness and related scale formation. Tests conducted at Purdue University found ....no significant, beneficial variation in the physical or chemical water quality parameters measured.
Items to consider when purchasing an ion exchange water softener
Test your water to determine the hardness and other impurities that may need to be removed.
Determine how much softened water your household needs per day, per year.
What type and size of softener will fit your situation?
How easy is the softener to clean and/or repair?
What type of convenience level should a softener offer 'manual or automatic operation'?
Will pretreatment be needed for iron and manganese?
Will sodium intake be a health problem?
Will sodium salts overload your septic or sewer system?
Purchase price does not directly indicate a softener's performance. A moderately priced unit might work as well as an expensive unit.
Don't buy more equipment than you need. Other removal systems might be better suited for the removal of certain impurities.
Further Information
For further information contact your local county extension office or state health department.
References:
University of Nebraska Extension
Research Reports: Quantitative Assessment of the Effectiveness of Permanent Magnetic Water Conditioning Devices. Water Quality Association, 1985
Funding for this publication was by the U.S. Department of Agriculture, Extension Service, under project number 90-EWQI-19252.
AE-1031, February 1992
Original Article
http://www.ag.ndsu.edu/pubs/h2oqual/watsys/ae1031w.htm
| |
|
Images are representative of the products. Images may or may not be of the actual product. If it is important e-mail us for an actual image if available.
* Flat Rate UPS shipping when able to ship via UPS and is in the USA excluding Hawaii and Alaska.
Larger Items may not be able to ship via UPS, in that case freight charges will be quoted seperately.
International shipping will be quoted after the order is placed. You will have the opportunity to cancel before we finalize your order.
Terms and conditions
Credit Application
Privacy
Policy
List All Products
|
















